Pharmaceutical companies struggle with costs and complexity in Phase III
Before they can go to market, drugs must pass through a series of rigorous trials. Phase III trials, in which drugs are tested on human subjects, take several times as much time and money than Phase I and II, costing between $1.5 and $2 billion for a single drug.
Clinical trails currently cost between $1.5 and $2 billion for testing a single new drug.
A simple way to improve the process would be to build predictive models to better identify patients whose symptoms could be exacerbated by the trial drug. This approach could save billions of dollars and result in more effective drugs reaching the market. Unfortunately, there are problems with this method:
- Data analysis is usually biased by whoever performs it.
- To ensure accuracy, other independently derived solutions need to be matched against the one being tested.
- Patient data is subject to strict HIPAA laws and regulations that make it difficult to work with.
Pharmaceutical companies have rightfully been cautious to implement this approach. How could drug manufacturers take advantage of AI solutions without compromising on Phase III trial effectiveness or patient security?
A combination of effectiveness and efficiency
An example of an effective use of this method would be in the identification of COPD (chronic pulmonary obstructive disease) as a risk factor for clinical trial participants moving past Phase II. If a wide variety of patient data could be collected, models could be developed that would predict the likelihood of a patients’ COPD becoming a drug trial risk factor. This means not only data such as patient survey responses and baseline health data, but also their demographic, clinical, imaging, and genetic information.
Phase III trials could be more accurately targeted with specific patient profiles, resulting in more accurate Phase III trial results.
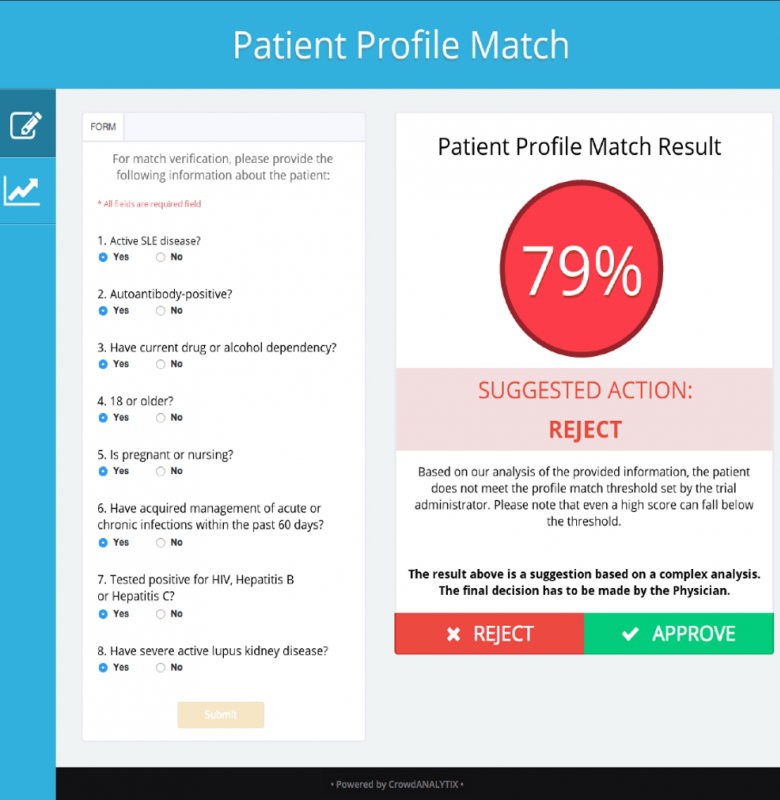
By combining this data with longitudinal follow-up outcomes, data scientists could develop an algorithm to predict which patients are more likely to exhibit an exacerbation than others and uncover the top variables which could help identify and monitor exacerbation of disease. This way, Phase III trials could be more accurately targeted with specific patient profiles, resulting in more accurate Phase III trial results.
Our approach
By using a widespread, crowdsourced team of data scientists, CrowdANALYTIX was able to eliminate the biases that can make data analysis less accurate. Additionally, by allowing dozens of data scientists access to the same data, all fully anonymized for HIPAA compliance, CrowdANALYTIX produced the multiple independently derived approaches necessary for determining the accuracy of each solution. The high number of independently produced solutions also compensates for any gaps in data. In all, CrowdANALYTIX received 1,232 submissions from a group of more than 300 data scientists.
Because these artificial intelligence solutions accounted for almost 1500 variables, including patients’ genetic data, they reached a very high level of accuracy in determining patient outcomes in Phase III trials.
Impact
Based on the Phase III selection criteria derived from CrowdANALYTIX’ predictive model, the client was able to perform a statistically valid trial with at least 15% fewer patients and run safer trials by utilizing the predictive model to identify specific patients that may need greater observation or care during the trial.



